Many undergraduates that have been introduced to NMR understand the concepts of chemical shift and coupling, however struggle when presented with a real 1H NMR spectrum. For a novice at interpreting proton NMR spectra, it can be difficult to distinguish between separate resonances and multiplets belonging to the same resonance.
‘JRES’ is a 2D homonuclear experiment that produces a J-resolved spectrum, i.e. the chemical shift along one axis (f2) and the proton-proton coupling along the other axis (f1). Essentially, the projection along f2 is a decoupled proton spectrum, which greatly simplifies the spectrum and allows students to quickly identify the chemical shift and coupling pattern.
Comparison of the JRES spectrum with the 1D proton spectrum allows students to understand how proton-proton coupling affects the appearance of the spectrum and become more confident at distinguishing between multiplets and different resonances.
Example: Ethyl crotonate
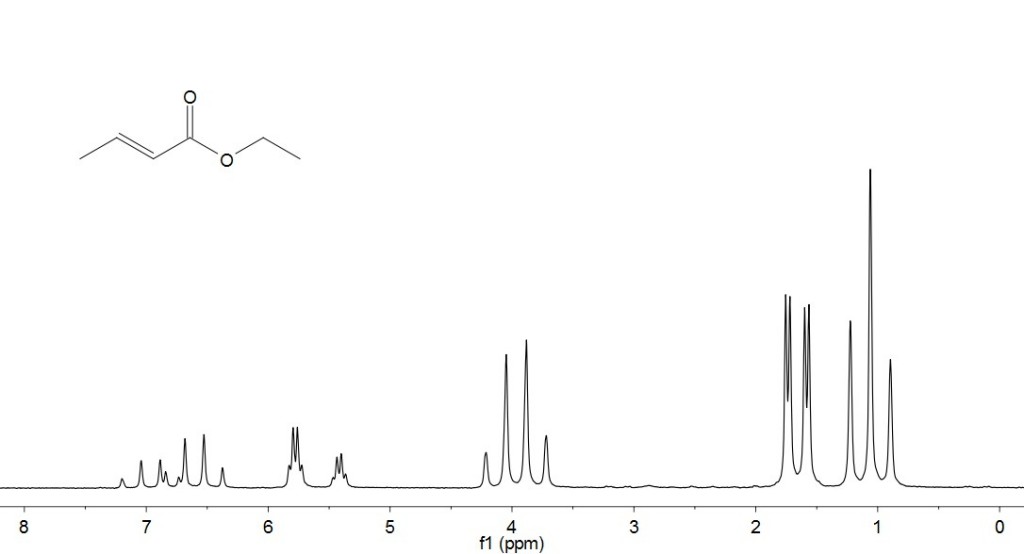
The assigned spectrum can be found here.
In the 1D proton NMR spectrum there are a set of peaks around 5.5-6 ppm. It is not obvious to the beginner whether these are two separate resonances or a large doublet. In the JRES we can see that in fact the two resonances collapse into one peak in the f2 dimension and is split into a doublet in the f1 dimension, which means the peaks are one proton split into a doublet. From the 1D proton spectrum it is also difficult to determine whether the set of peaks between 6.3-7.2 ppm is one or multiple resonances, and what the splitting pattern is. This is resolved in the JRES spectrum where again all of the peaks collapse into one in the f2 dimension, so the set of peaks is due to one resonance. It can also be determined by JRES that it is split into a doublet of quartets, which is difficult to see in the 1D proton NMR spectrum.
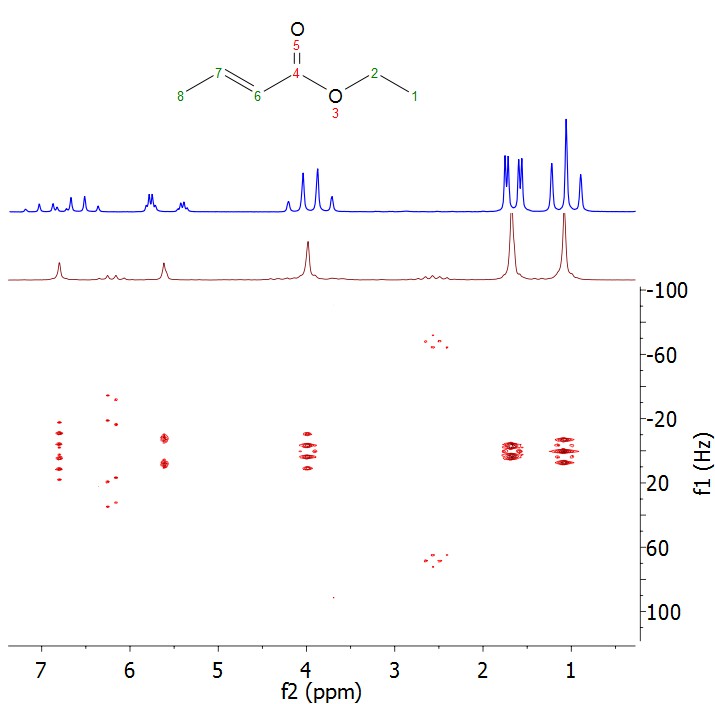
The assigned spectrum can be found here.
We have developed an Application Note using JRES to deconvolute the spectra of aromatic compounds, which can be found here.