680. Impact of the Anion Structure on Coordination and Dynamics in a Localized High-Concentration Battery Electrolyte
Anne Hockmann, Peng Yan, Diddo Diddens, Isidora Cekic-Laskovic, Monika Schönhoff, JPhysChemB, (2025), DOI: 10.1021/acs.jpcb.5c01566
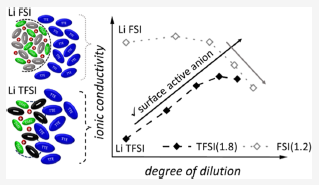
Localized high-concentration electrolytes (LHCEs) are based on the immiscibility of a concentrated salt phase and a diluent phase, creating an internal interface. Here, we study LHCEs based on lithium bis- (fluorosulfonyl)imide or lithium bis(trifluoromethanesulfonyl)imide as the conducting salt, 1,2-dimethoxyethane as the solvent, and 1,1,2,2-tetrafluoroethyl- 2,2,3,3-tetrafluoropropyl ether (TTE) as the diluent. With increasing dilution, trends of dynamic properties such as ionic conductivity and self-diffusion strongly depend on the anion structure. By analyzing ion coordinations by NMR and Raman spectroscopy, we attributed this to a different interfacial affinity of the anions: TFSI− anions are more likely than FSI− anions to interact with the diluent molecules. This stabilizes the TTE| conducting salt interface, causing a less compact and more mobile conducting salt-rich phase. Furthermore, it leads to a higher degree of ion dissociation, which explains the enhanced molar ionic conductivity found upon dilution of TFSI-based LHCEs, as opposed to FSI-based LHCEs. These differences are most probably due to the larger size and enhanced charge delocalization of the TFSI− anion compared to the FSI− anion, making the interaction with the uncharged TTE molecule more favorable. Thus, understanding local ion coordinations in different electrolyte formulations plays a crucial role in optimizing transport properties.