747. Participation of the Cyanide Group in the Reaction Mechanism of Benzoxazole Formation: Monitoring by Continuous Flow Cell NMR
Nelda Xanath Martínez-Galero, Daniel Galindo, Lemuel Pérez-Picaso, Lucio Peña-Zarate, ChemistryProceedings, (2025), 10.3390/xxxxx
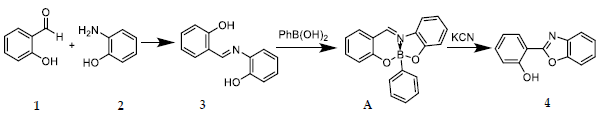
Benzoxazoles are recognized as significant building blocks in organic synthesis and materials science. This work observed the formation of benzoxazole from o-aminophenol and ohydroxybenzaldehyde using online 1H NMR (continuous flow cell, 80 MHz). The identification of changes in the functional group was complemented by ATR-FTIR analysis. Additionally, the kinetic roles of phenylboronic acid and cyanide in the one-pot condensation-cyclization reaction are examined. Real-time monitoring has revealed three observable events: the rapid condensation of the aldehyde and o-aminophenol to produce the imine; the formation of the boron complex in the presence of phenylboronic acid; and the cyanide-assisted cyclization that converts the intermediate into benzoxazole. The findings clarify the transformations that control throughput and provide valuable insights for optimizing reagent loadings under environmentally friendly conditions.