770. Transport and structural properties of a water-in-Bisalt electrolyte: LiTFSI and EmimTFSI mixtures
Horacio R. Corti, Facundo C. Herrera, Maximiliano Drelewicz, Santiago Drelewicz, Paula Y. Steinberg, Rodolfo H. Acosta, Fernando Pschunder, Lucas M. Montenegro, JMolLiquids, (2025), 10.1016/j.molliq.2025.129186
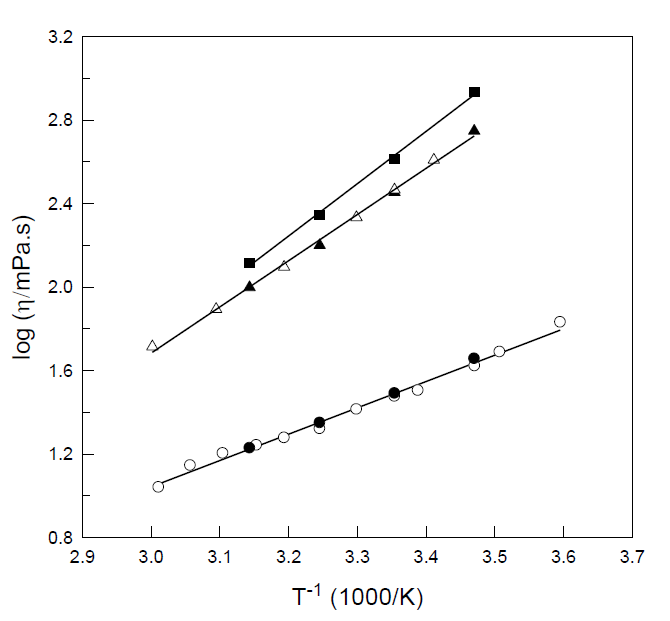
Water-in-Salt electrolytes, ionic liquids, and their mixtures have been proposed as electrolytes for advanced batteries and supercapacitors. In this work, we studied the volumetric, transport properties (viscosity, electrical conductivity, diffusivity), and nanostructure of two Water-in-Bisalt electrolytes (WiBE) formed by the ionic liquid, 1-ethyl-3-methylimidazolium bis(trifluorometha- nesulfonyl) imide ([Emim][TFSI]), and lithium bis(trifluoromethanesulfonyl) imide (LiTFSI), at temperatures between 288.15 and 318.15 K. We observed that, as found for the aqueous binary systems LiTFSI-water and [Emim][TFSI]-water, the ternary system LiTFSI-[Emim][TFSI]-water behave as ideal mixtures regarding their volumetric properties. A conductivity/viscosity decoupling is observed that can be described by a fractional Walden law with decoupling coefficients close to 0.85. The diffusivity of the species determined using diffusion-ordered (DOSY) NMR spectroscopy of 1H, 19F and 7Li, follows the order H2O > Emin+ > Li+ > TFSI- and the apparent transport number of the Li+ ion reached 0.44. The nanostructure of the WiBEs, studied by Small-Angle X-Ray Scattering (SAXS), shows two peaks at high scattering vector (q), previously predicted by Molecular Dynamics simulations, whose shift with increasing concentration of LiTFSI is related to the formation of TFSI- anion chains, a fact that correlates with its low diffusivity. Complementary, Small-Angle Neutron Scattering (SANS) reveals the mesoscale structure described by the Teubner-Stray model, with periodicity distances close to 1 nm, slightly lower than that observed for LiTFSI-based Water-in-Salt electrolytes (WiSE) with lower salt LiTFSI concentration.