786. Expanding the Range of Methods for Obtaining Diverse Representatives of Sulfonyl Phosphoramidate Oligonucleotides
Sergey A. Zhukov, Ekaterina G. Seroklinova, Maxim S. Kupryushkin, ACSOmega, (2026), 10.1021/acsomega.5c11209
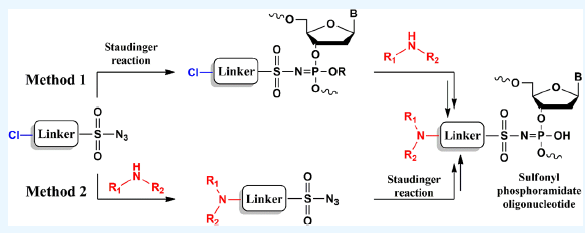
Sulfonyl phosphoramidates are a class of internucleotide phosphate modifications that can be easily introduced during automatic solid-phase synthesis at the oxidation step using the Staudinger reaction with appropriate electron-deficient sulfonyl azides. Beyond altering the nature of the backbone, this class of modifications enables the introduction of various substituents within the structure of oligonucleotides. The existing method, which relies on a limited set of precursors, enables obtaining only a narrow range of representatives within this class. In this work, we report an approach for obtaining diverse representatives of the sulfonyl phosphoramidate class based on the introduction of commercially available building blocks with different functional groups into the structure of modification. The first proposed method involves the incorporation of chloroalkanesulfonyl azides, followed by the substitution of the chlorine atom with amine residues within the sulfonyl phosphoramidate derivative. Initial evaluation of a series of sulfonyl azides bearing terminal chlorine atoms on alkyl chains of varying lengths (C1−C3) led to the selection of 3-chloropropanesulfonyl azide as the most promising agent. A library of sulfonyl phosphoramidate derivatives bearing various amine residues, including bulky alkyl and polyamine moieties, was synthesized by using this azide. The introduction of reactive groups into a growing oligonucleotide chain enabled the multistep assembly of complex structures on a solid-phase support. In addition, the second method was proposed, involving the synthesis of sulfonyl azides already bearing amine residues for subsequent incorporation within oligonucleotides. This method can be implemented via three synthetic routes: introducing a lipophilic amine residue into the chloroalkanesulfonyl azide structure, synthesis of azides via sulfolactone ring-opening alkylation to obtain quaternized derivatives, and obtaining sulfamoyl azides. The developed methods are complementary, with one enabling the effective incorporation of functional residues that are difficult to introduce with the other two. The proposed approach enables a significant expansion of the set of sulfonyl phosphoramidate oligonucleotide derivatives by utilizing commercially available building blocks and straightforward reactions.