801. From Glycerol to Xylitol: Chain-length Effects and Selectivity in Alkaline Polyol Electrooxidation
David Tran, Zhe Meng, Fabian Luca Buchauer, Amelia Rose, Henrik H Kristoffersen, Jan Rossmeis, Johan Hjelm, Chemrxiv, (2026), 10.26434/chemrxiv.15000838/v1
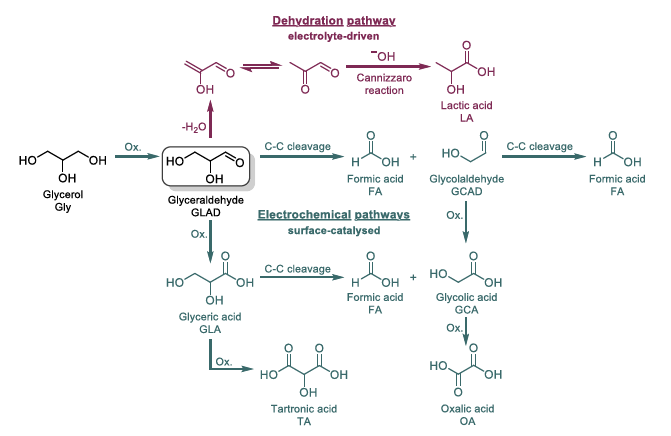
Glycerol electro-oxidation is a promising way to utilise the oversupply from biodiesel production and simultaneously produce valuable organic acids as well as H2. Another promising bio-derived polyol for commodity chemicals production is xylitol, yet it has not attracted as much scientific attention as glycerol. The extensive studies on glycerol provide a good foundation for evaluating the reaction pathways of xylitol and for identifying molecular-size-dependent differences. In this study, we systematically investigate the influence of the polyol chain lengths of glycerol, erythritol, and xylitol on the electrooxidation and product distribution. We contrast the outcomes of using Pt and Ag electrodes for the anodic oxidation of these polyols in alkaline medium, as computational studies have shown a preference for carbonyl-C* adsorption on Pt and hydroxyl-O* adsorption on Ag, leading to different reaction paths and additional length-dependent effects. On Pt, we first observe a single oxidation process to the corresponding aldehyde. Subsequently, homogeneous CC bond cleavage of erythrose and xylose in the alkaline reaction media dominates the product outcome, while glyceraldehyde mainly yields C3 acids upon further oxidation. For Ag, we observe the formation of the same three acids, glyceric-, glycolic and formic acid. Our findings indicate that direct CC cleavage occurs alongside oxidation, yielding one acid and one aldehyde in the first reaction step. Dependent on the polyol length, the ratios of the three resulting acids vary, providing valuable information about the preferred splitting behaviour. This study provides important insights into the electrooxidation of larger polyols than glycerol, with bearing on their suitability for selective chemicals production.