735. Assessment of edible essential oils protection on the thiyl radical cis–trans isomerization of methyl oleate
Styliani Chatziprokopiou, Michael A. Terzidis, JFoodCompAnalysis, (2025), 10.1016/j.jfca.2025.108531
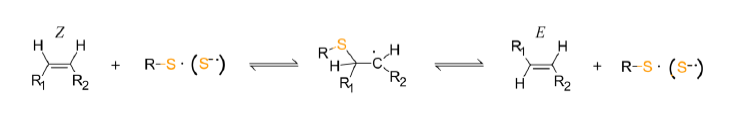
The influence of i) rosemary, ii) lavender, iii) sage and iv) peppermint essential oils on the thiyl radical methyl oleate cis–trans isomerization efficiency was investigated. To that end, some commonly used antioxidants (trolox and ascorbic acid) were also explored along with linalool, eugenol and two terminal alkenes (4-methylstyrene and 1,1-diphenylethylene). The role of the thiyl radical origin to the cis–trans isomerization was examined by using different thiols, namely a) 2-mercaptoethanol, b) N-acetylcysteine and c) 2,2-dithiothreitol, also d) dithioether 5,5-dithio-bis-(2-nitrobenzoic acid). All the essential oils screened found capable to inhibit the trans formation but the inhibition effect found dependent on the chemical characteristics of the thiyl radical and the essential oil composition. Rosemary oil containing large amounts of terminal alkenes gave the best thiyl radical scavenging results followed by sage, lavender and peppermint while from the pure compounds studied, 4-methylstyrene and 1,1-diphenylethylene provided the highest protection effect.