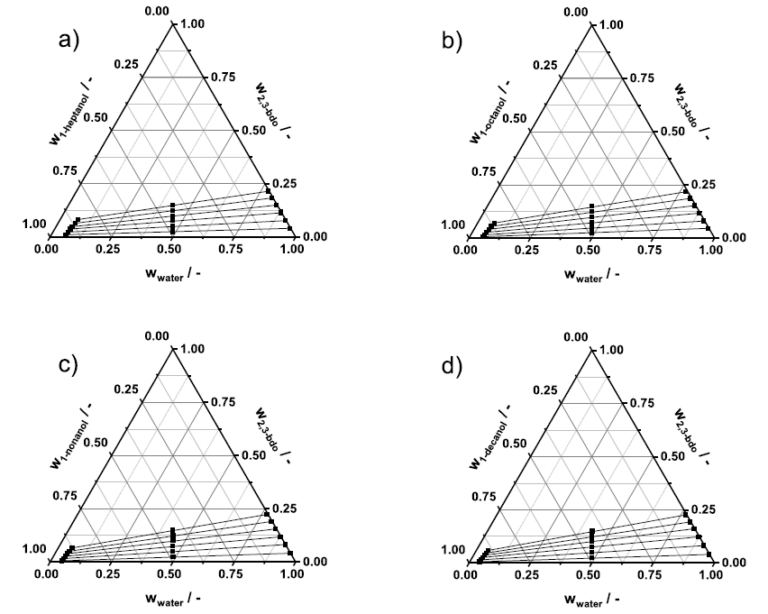
769. Liquid-liquid equilibrium of water + 2,3-butanediol + n-alcohols (1-heptanol, 1-octanol, 1-nonanol, 1-decanol) and water + acetoin + n-alcohols ternary systems at 298.2 K
Peter Forster, William Graf von Westarp, Janik Hense, Johannes von Campenhausen, Andreas Jupke, FluidPhaseEquilibria, (2025), 10.1016/j.fluid.2025.114657
Liquid-liquid equilibrium (LLE) data were experimentally determined for ternary systems containing 2,3-butanediol (2,3-bdo) or acetoin as solutes, water and 1-heptanol, 1-octanol, 1-nonanol, and 1-decanol as solvents, at 298.2 K and atmospheric pressure. Distribution coefficients increased with decreasing carbon-chain length of the solvents, ranging from 0.2610 (1-decanol) to 0.3827 (1-heptanol) for 2,3-bdo and from 0.2817 (1-decanol) to 0.4252 (1-heptanol) for acetoin. Acetoin exhibited higher maximum distribution coefficients (0.4252 in 1-heptanol) and selectivities (7.9429 in 1-decanol) compared to 2,3-bdo (0.3827 in 1-heptanol and 4.5745 in 1-heptanol, respectively). Trends are consistent with solute functionality and solvent polarity. Experimental LLE data were correlated using the NRTL activity coefficient model, and the binary interaction parameters were successfully regressed. A topological analysis confirmed the consistency of the model correlations at 298.2 K.