783. Synthesis, Characterization, Optical Band Gaps of a Zn(II) Schiff Base Complex and Application in Photocatalytic Degradation of Organic Dyes
Fatema Zohra Chiboub Fellah, Abdelghani Chiboub Fellah, Soumia Boulefred, Racha Bouchenak Khelladi, ChemistryAfrica, (2026), 10.1007/s42250-025-01564-x
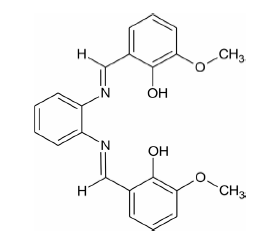
In this study, a zinc (II) complex was synthesized with a Schiff base ligand derived from orthovanillin and ortho-phenylenediamine. The formation of the obtained complex and the coordination of the ligand to the metal center were confirmed by various spectroscopic techniques, including infrared (IR), ultraviolet-visible (UV-Vis), thermogravimetric analysis (TGA), and nuclear magnetic resonance (NMR).The optical band gap of the complex, determined via UV-Vis spectroscopy and Tauc analysis, reveals a direct transition at 3.75 eV, confirming its semiconducting nature. The photocatalytic activity of the complex was evaluated under UV irradiation for the degradation of two widely used organic dyes: methylene blue (MB) and crystal violet (CV). Several parameters were studied to optimize the degradation efficiency, including the mass of the photocatalyst, the volume of hydrogen peroxide (H2O2), and the effect of sunlight. Results revealed that the complex exhibits excellent photocatalytic efficiency, particularly under UV light in the presence of hydrogen peroxide, with degradation rates exceeding 95% for both dyes under optimal conditions (catalysts mass = 4.5 mg, H2O2 volume = 4.4 mL for MB and 5.4 mL for CV). Moreover, the study highlights the potential of this Zn-based catalyst for environmental applications, offering an effective and sustainable approach for treating dye-contaminated wastewater.