789. Integrated spectral, structural and computational investigation of antihypertensive drug Clonidine: experimental, DFT and molecular interaction analysis
Suganthi Manickam, Sambasivam Ragavan, Ramalingam Singaravelu, Tarun Yadav, Vetrivelan Vaithiyananthan, ChemicalPapers, (2026), 10.1007/s11696-026-04640-9
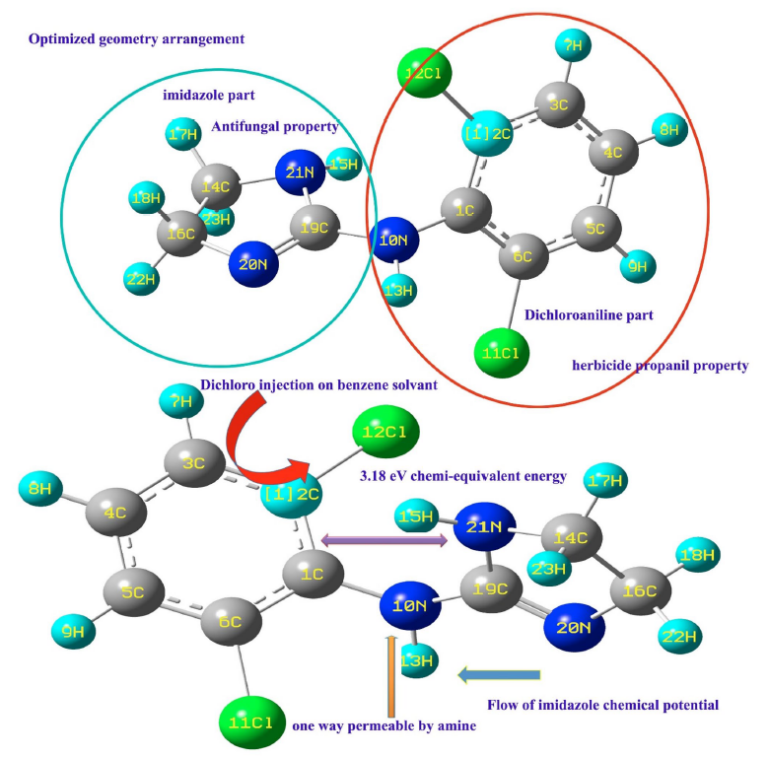
Clonidine, an established antihypertensive drug, was investigated through integrated experimental (FT-IR, FT-Raman, NMR, UV–vis) and computational (DFT/B3LYP) analyses to elucidate its structural, vibrational, electronic, and molecular interaction characteristics. Experimental spectra were correlated with scaled DFT frequencies, confirming key functional group vibrations associated with the imidazoline and dichlorophenyl units. The optimized geometry showed good agreement with the reported single-crystal XRD structure (CCDC 185578). The HOMO–LUMO energy gap (4.58 eV) indicates molecular stability with limited intramolecular charge transfer. NMR chemical shifts were rationalized using computed shielding tensors, highlighting the influence of electronegative chlorine and ring-current effects. UV–vis spectra revealed π→π* and n→π* transitions consistent with the electronic configuration of the molecule. ADME profiling predicts acceptable drug-likeness and pharmacokinetic properties. Molecular docking with the 5UUT protein indicates moderate binding affinity (– 6.59 kcal/mol) via hydrogen bonding and hydrophobic interactions, consistent with its known receptor interactions. The combined spectroscopic and theoretical results provide a validated molecular description of Clonidine and reinforce its structural stability, electronic features, and pharmacokinetic suitability without claiming new therapeutic indications.