799. Cyclometalated Platinum Compounds from Competing C−H/C−X Bond Activation Pathways
Craig M. Anderson, Matthew W. Greenberg, Christopher N. LaFratta, Monika Dziubelski, Zainab Aleem, Benett B. Hathaway, Joseph M. Tanski, ACSChemicalBiology, (2026), 10.1021/acsomega.5c12884
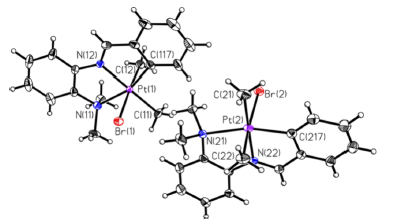
X−C^N^N (X = Br, Cl) ligands were reacted with [Pt2Me4(μ-SMe2)2], 1, resulting in a six-coordinate cyclometalated platinum(IV) compound containing an anionic C^N^N ligand when X = Br and both a platinum(IV) and a platinum(II) product when X = Cl. The platinum(II) species was formed by C−H activation, followed by reductive elimination of methane. The platinum compounds were characterized by multinuclear NMR spectroscopy and single-crystal X-ray diffraction (SCXRD). Photophysical properties were explored by using UV/vis, emission, and transient absorption (TA) spectroscopies. DFT and TDDFT calculations were performed to examine the competition between C−H activation and C−X oxidative addition and compared to experimental results.