808. Glycolytic alterations as biomarkers in polycystic kidney disease: A study using a PKD1 knockout model in NRK-52E rat kidney epithelial cells
Ida Kjær Mieritz, Christoffer Laustsen, Esben Axelgaard, Sofie Rahbek Dorset, Rasmus O. Bak, Lotte Bonde Bertelsen, PhysiologicalReports, (2026), 10.14814/phy2.70816
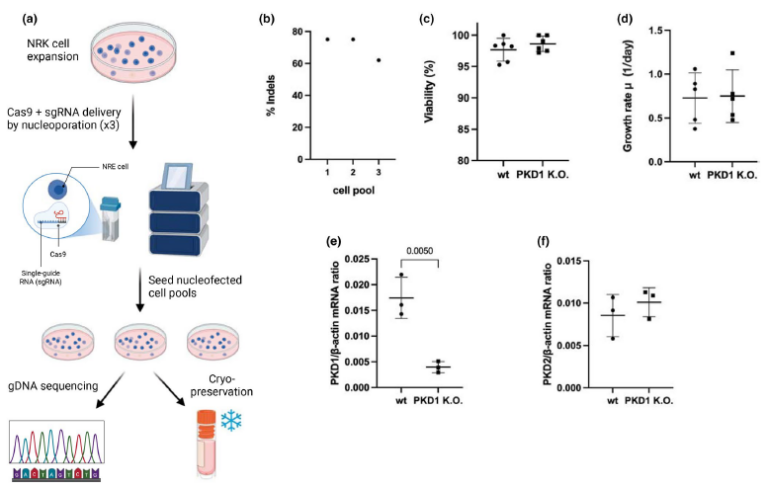
Polycystic kidney disease (PKD) is a genetic disorder characterized by the formation of fluid-filled cysts in the kidneys, often resulting in progressive renal impairment. Mutations in the PKD1 gene represent the predominant genetic cause of autosomal dominant PKD. Here, we investigated how PKD1 knockout affects glycolytic metabolism in NRK-52E kidney epithelial cells using dynamic nuclear polarization (DNP)-enhanced magnetic resonance spectroscopy (MRS) with hyperpolarized [1-13C] pyruvate. PKD1 knockout NRK-52E kidney epithelial cells showed a significantly elevated pyruvate-to- lactate conversion as measured by hyperpolarized [1-13C] pyruvate (HP-MRS) and significantly increased lactate levels in culture medium, accompanied by upregulated lactate dehydrogenase (LDH) gene expression and enzymatic activity. Monocarboxylate transporter (MCT) expression was selectively altered (significant downregulation of MCT2 and MCT3; MCT1 not significantly changed). Pyruvate dehydrogenase (PDH) activity and transcript levels did not differ between groups. These results demonstrate glycolytic reprogramming associated with PKD1 deficiency and support hyperpolarized pyruvate MRS as a sensitive metabolic biomarker for detecting such alterations in real-time. These findings identify glycolytic remodeling as a robust metabolic consequence of PKD1 loss and demonstrate that HP-[ 1-13C] pyruvate MRS provides a flux-level biomarker suitable for real-time metabolic characterization.