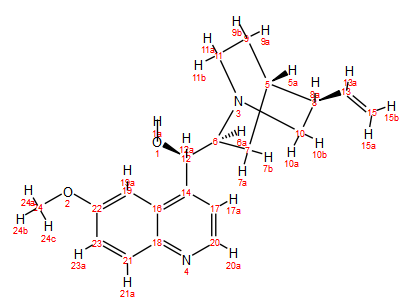
In my first post on using 1D and 2D NMR methods to assign the peaks of quinine (Figure 1), I looked at the 1H spectrum.
Figure 1. Structure of quinine
In this post, I’m moving on to look at the 13C spectrum. Figure 2 shows the 13C spectrum of the 400 mM quinine sample used in this study.
Figure 2. 13C NMR spectrum of 400 mM quinine. The spectrum was collected using 512 scans in 26 minutes.
In the 13C spectrum, 17 peaks can be clearly observed (excluding the septet signal from the DMSO-d6 solvent centered at δ = 39.5 ppm). The intense peak at 27.6 ppm is likely to be due to two carbons that have the same chemical shift, which brings the carbon signal count to 18, two short of the 20 carbon signals we expect from quinine. It’s worth remembering, however, that there may be some additional peaks buried beneath the solvent signal. In terms of peak assignments, based on its chemical shift, the downfield signal at δ = 157.3 ppm can be assigned to C22. Other than that, it’s difficult to assign with any degree of certainty the other peaks in the spectrum. The assignments made so far using the 1H and 13C spectra are shown in Table 1.
Atom |
1H (ppm) |
Peak Splitting Pattern |
J (Hz) |
13C (ppm) |
24 |
3.55 | — | — | |
| 20 | 8.33 | d | 4.5 | |
| 21 | 7.59 | d | 9.1 | |
| 23 | 7.03 | dd | 9.1, 2.7 | |
| 15 (tent.) | 4.6 | m | — | |
| 22 | — | — | — | 157.3 |
Table 1. Preliminary 1H and 13C peak assignments obtained from the 1D spectra
To make the remaining assignments we will need to use 2D methods. That will be the subject of my next post.