Monitoring Chemical Reactions Performed at Temperatures up to 60 °C by NMR
NMR is an invaluable tool for real-time monitoring of chemical reactions, with capabilities including but not limited to the measurement of percent conversion, kinetic studies, side product formation, and global reaction optimization1,2,3,4. Driven by a collaborative project between Magritek and process chemists from major pharmaceutical companies, as a part of an Emerging Technologies Consortium project on the development of a Portable NMR Reaction Monitoring Platform, we have developed the new Spinsolve Sample Temperature Control system (STC) which allows for online measurements of the reaction mixture from room temperature up to 60 °C.
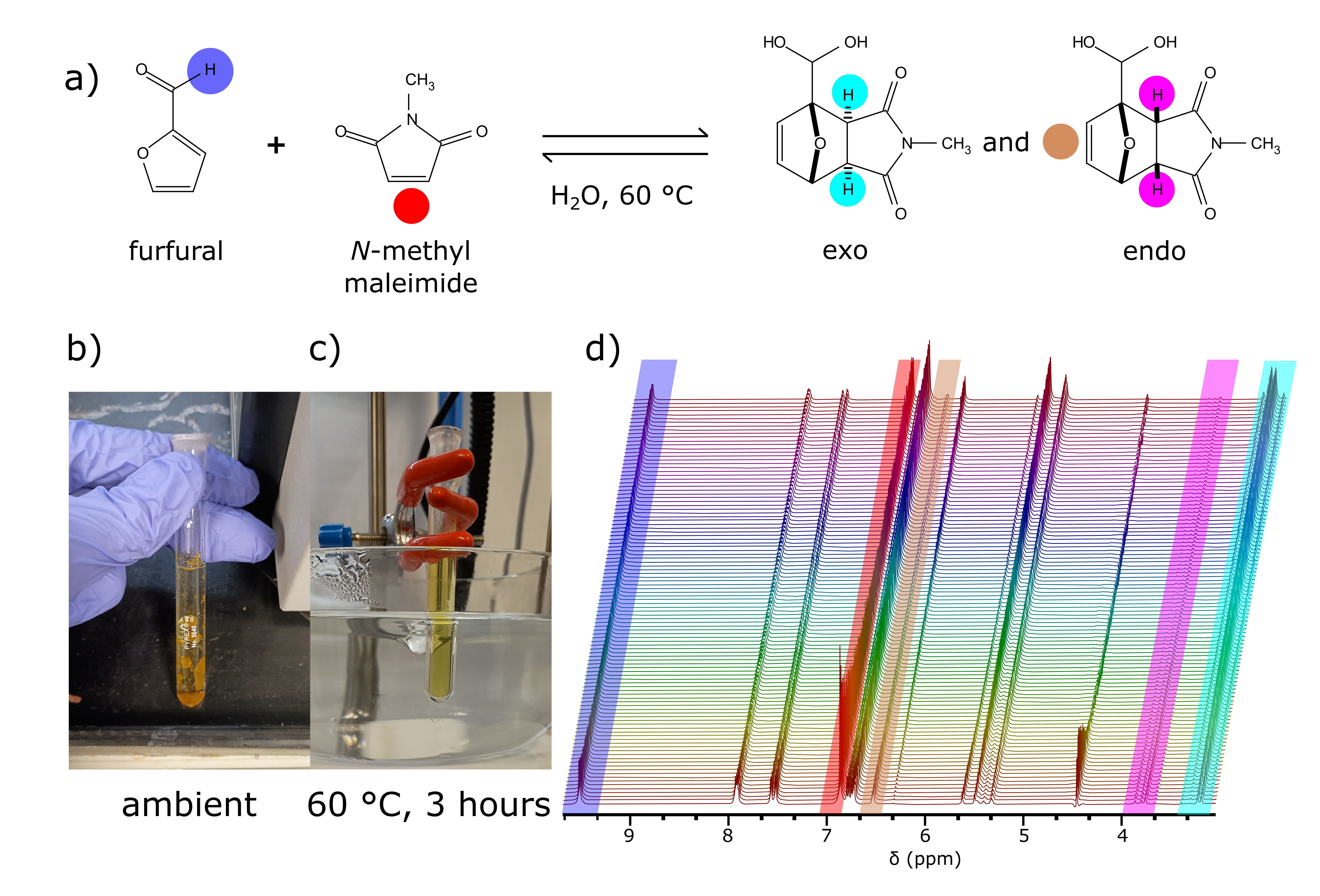
Figure 1. a) Diels-Alder reaction scheme. b) At room temperature the reactants are insoluble. c) After extensive heating, the solution becomes homogenous. d) Stack of solvent suppressed 1H data from the Diels-Alder reaction. The signals of interest are color coded to match the circles in a).
Industrial chemical processes are commonly run at elevated temperature due to solubility or reactivity considerations; therefore, room temperature NMR measurements are applicable to a limited reaction scope. To demonstrate the necessity of sample temperature control technology to study certain reactions with benchtop NMR, in this blog post we monitor, in an NMR tube, a Diels-Alder reaction (DA) between N-methyl maleimide and furfural based on the approach of Cioc R. et al 5 (see the reaction scheme in Figure 1a). In Figure 1b and 1c, we show that the reagents are insoluble at ambient temperature, while at 60 °C the mixture becomes homogenous.
NMR Results & Discussion
Representative 1D spectra at 5, 13, and 139 hours are shown in Figure 2. Note that in all spectra, the residual water signal is approximately 20 Hz, 0.25 ppm in width, which is exceptionally narrow while at 60°C. Since there are multiple resolved signals for each reaction component, it was straightforward to track the reaction progress in a similar manner to the reference paper’s high field NMR measurements5.
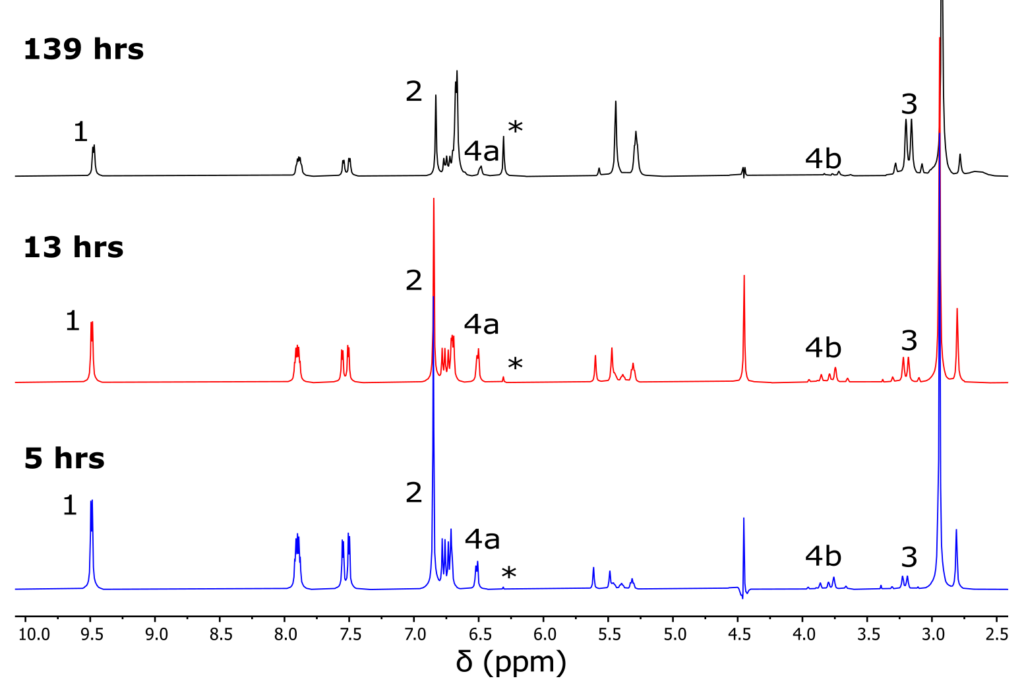
Figure 2. Three representative 1D 1H spectra with WET suppression and 4 scans at the indicated reaction time. The labeled peaks are used to track reaction species concentrations over time (see Figure 3). 1 is furfural, 2 is maleimide, 3 is exo product, 4a and 4b are endo product peaks, and the * is a side product. Note that the width of the baseline of the residual water peak is less than 20Hz.
The reaction produces a mixture of endo (minor) and exo (major) products. Percent yield of endo, exo, and total product are shown in Figure 3a. The endo product peaks between 4-3.5 ppm are not attenuated by the WET module applied to suppress the water signal only 0.5 ppm away, since the percent-yield calculated is the same as when using the alternative endo peak at 6.5 ppm, Figure 3a. Thus, fully protonated solvents can be used even at elevated temperatures while the Spinsolve retains excellent solvent suppression performance.
By plotting the percentage of reactant consumed in Figure 3b, we see that maleimide and furfural react at the expected ratio of 1:1. The deviation at the final time point for each reactant consumed, 694 vs 722 mM, 4% different, might be caused by side-product formation. Indeed, in Figure 2, we see a side product forming near 6.3 ppm, especially at later time points. In Figure 3c, the diastereomer excess is examined; clearly the endo product is the kinetic product, while the exo product is the thermodynamically favored product. The final diastereomeric excess is measured to be 78%.
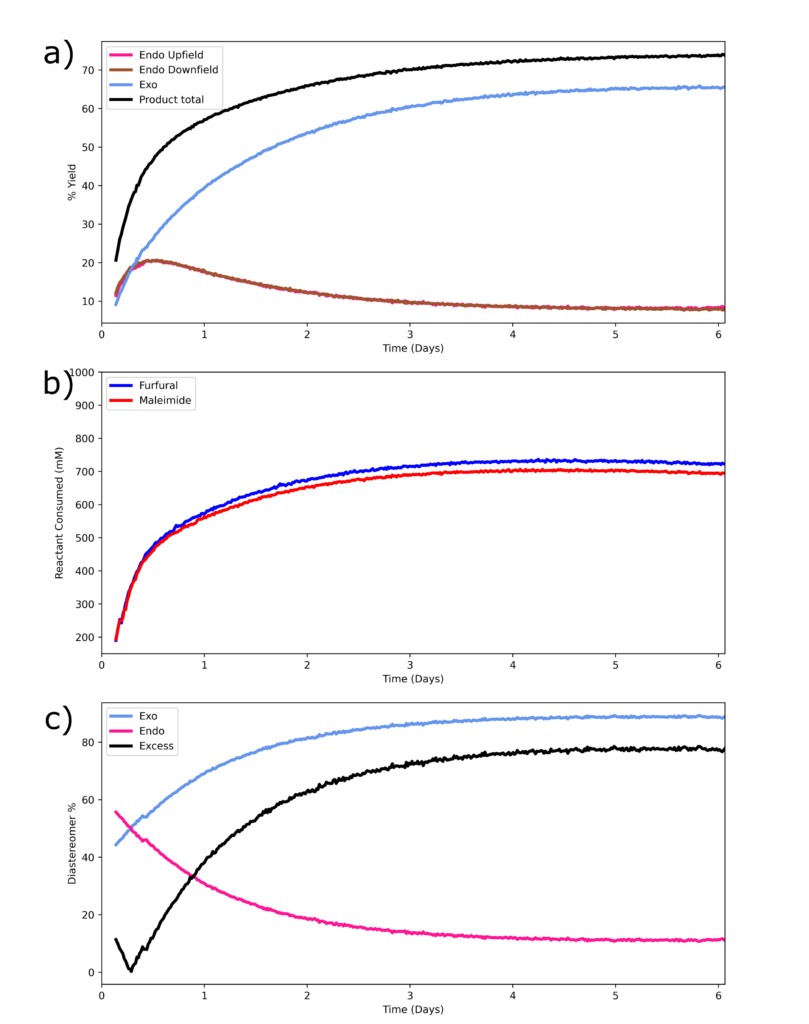
Figure 3. a) % yield of the products; this is calculated with respect to the initial maleimide concentration, the limiting reagent. The two endo datasets show the same result and overlap in the graph. b) Reactant consumed over the course of the reaction. The initial reaction spectrum was used to calibrate the NMR signal response to the known concentration, i.e. the integration sum for the exo, endo, and maleimide peak is equivalent to the mass-based initial maleimide concentration since there is negligible side reaction product at the initial time point. c) % of each diastereomer, and diastereomeric excess.
In the reference, the authors stopped the reaction at 60 hours while here the reaction was ran for six days. We see slightly higher % conversion, 73% relative to maleimide versus the reported 60-65%. We also measure slightly higher diastereomeric excess, 78 versus 67%, likely at the expense of reduced product purity, as side-product-related NMR peaks, such as at 6.3ppm in Figure 2, are observed to continue to increase at longer time points5.
Conclusion
A room-temperature insoluble Diels-Alder reaction was studied in fully protonated solvent at 60 °C using the Spinsolve 80 ULTRA STC spectrometer. Product interconversion and impurity formation were both continuously tracked over six days. ULTRA performance at elevated temperature was demonstrated by the lack of
attenuation of the endo-product peak despite the nearby suppressed water signal. This case study demonstrates how the Spinsolve’s Sample Temperature Control capability expands the scope of reactions able to be monitored in real time. Stay tuned for an upcoming post that will demonstrate the Spinsolve’s STC capability as an online/inline PAT tool for elevated temperature reactions when coupled with Magritek’s Reaction Monitoring Kit.
References
1. Maschmeyer, T. et. al. MRC vol. 64-4, 310-322 (2023). DOI: 10.1002/mrc.5395
2. https://magritek.com/2024/08/14/transesterification-of-pinacol-arylboronic-ester-how-to-monitor-heterogeneous-reactions-online-by-benchtop-nmr/
3. https://magritek.com/2025/06/13/automated-optimization-of-a-flow-reactor-by-using-bayesian-algorithms-and-inline-nmr-monitoring/
4. https://magritek.com/2023/03/13/monitoring-the-oxidation-of-phosphine-ligands-using-31p-nmr/
5. Cioc, R. et. al. Green Chem. 23, 367 (2021). DOI: 10.1039/d0gc03558k
To read the complete App Note please Click Below