708. A one-pot strategy for the synthesis and functionalization of hyperbranched polytriazoles
Hyeongju Noh , Jaehyeon Kim , Jina Min , Se Yeon Cheon and Si Kyung Yang, Designed Monomers and Polymers(2025), DOI: 10.1080/15685551.2025.2547342
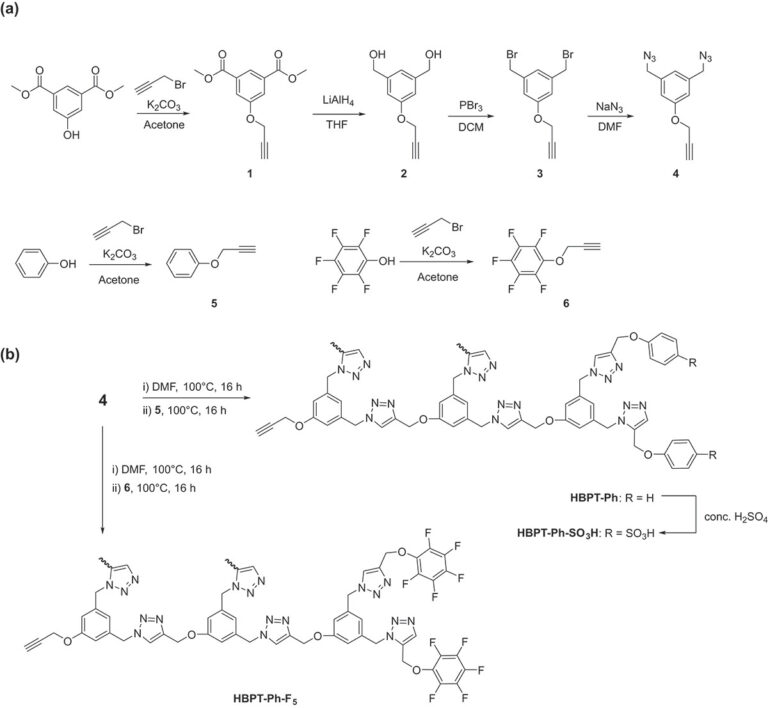
Herein we report a one-pot strategy for the synthesis and functionalization of hyperbranched polytriazoles by means of the Huisgen 1,3-dipolar cycloaddition of an AB2-type monomer containing an alkyne and two azide groups. The AB2 monomer is synthesized starting from dimethyl 5-hydroxyisophthalate in four steps with an overall yield of 58%, and the synthesis and purification are straightforward. The synthesis of end-capped hyperbranched polytriazoles (HBPTs) can be achieved via the Huisgen cycloaddition of the AB2 monomer using only heat, followed by functionalization with either sulfonic acid (-SO3H) or pentafluorophenyl (-Ph-F5) end groups. The resulting functional hyperbranched polytriazoles, HBPT-Ph-SO3H and HBPT-Ph-F5, are characterized by 1H NMR and FT-IR spectroscopies as well as gel-permeation chromatography. The film is fabricated simply by blending the two polymers, and the ion exchange capacity and ion conductivity are measured.