767. Green and Efficient Synthesis of Benzylidenemalononitrile Derivatives and α(/em>-Aminonitriles Using a Bifunctional MOF Catalyst
Sarita Kumari, Babita Poonia, Jyoti, Anand Yadav, Bhavana, Lalita Kumari, Satish Kumar Awasthi, Anindita Chakraborty, Prakash Kanoo, ChemistryAnAsianJournal, (2025), 10.1002/asia.202500766
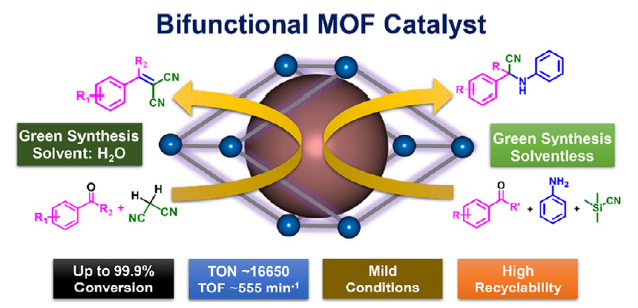
We report catalytic performance of a stable 3D metal–organic framework (MOF), {Mn2(1,4-bdc)2(DMF)2}n (Mn-BDC) (1,4-bdcH2,1,4-benzenedicarboxylic acid; DMF, N,N-dimethylformamide) in water and solventless condition. The coordinated DMF molecules in the MOF can be removed by heating to yield a desolvated phase, Mn-BDCDesolv with unsaturated Mn(II) sites (unsaturated metal sites [UMSs]) acting as Lewis acid sites, whereas the Lewis basicity arises from the carboxylate oxygens of 1,4-bdc. Mn-BDCDesolv is explored as a bifunctional heterogeneous catalyst to synthesize several benzylidenemalononitrile (BMN) derivatives in water and α-aminonitriles in solventless condition. The catalyst demonstrates excellent conversion (>99%) and high turnover number (TON) up to 16,500 and turnover frequency (TOF) up to 550 min−1. Remarkably, Mn-BDCDesolv maintained > 99% conversion over 24 cycles during the synthesis of BMN derivatives. The catalysts broad substrate scope, robust nature, superior TON, and TOF values compared to the benchmark MOFs and high recyclability highlights its potential for various Lewis acid–base-catalyzed organic transformations. It is important to mention that Mn-BDC can be prepared in gram scale in lab easily making the catalyst economical. Further, the catalytic process with Mn-BDC can be termed “green” as it allows the reactions to proceed either in water or in solventless condition.