768. A Subtle Twist: Hantzsch Ester-Controlled Enantiodivergence Driven by Weak Non-Covalent Interactions
Emanuele Cocco, Alessio Carioscia, Fabio Pesciaioli, Maurizio Prato, Giacomo Filippini, Massimiliano Aschi, Armando Carlone, Chemrxiv, (2025), 10.26434/chemrxiv-2025-9zwv9
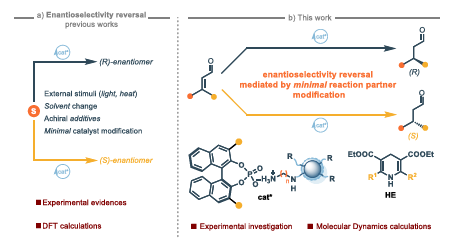
Enantiodivergence, the formation of opposite enantiomers from the same chiral catalyst, remains a largely empirical phenomenon in asymmetric catalysis, with limited mechanistic understanding. We report a remarkable case of enantiodivergence in the asymmetric transfer hydrogenation of α,β-unsaturated aldehydes, catalyzed by nitrogen-doped carbon dots (NCDs) and a fixed-handedness chiral phosphoric acid (CPA) under asymmetric counteranion-directed catalysis (ACDC) conditions. The stereochemical outcome is strongly influenced by minimal structural changes to the Hantzsch ester (HE) used as hydride donor. A synergistic investigation combining experimental results with extensive Molecular Dynamics (MD) simulations reveals the origin of this counterintuitive stereochemical switch. Our analysis show that the reversal in enantioinduction originates from differences in the reactive complex preorganization involving iminium ion, CPA, and HE. The simulations reveals that, under the experimental conditions, the preferred preorganization involves the CPA selectively coordinating to the iminium ion derived from the NCD–aldehyde adduct. This supramolecular assembly is steered by a delicate balance of weak London dispersion forces, which modulate the conformational ensemble of the catalytic system in solution and dictate the preferred face of hydride attack. This work provides a compelling rationalization for a complex phenomenon and underscores the critical role of subtle non-covalent interactions in controlling asymmetric reactions.