Long-Range 1H-31P HSQC for selecting one bond and multiple bond correlations
Since its introduction in the 1980s, heteronuclear two-dimensional (2D) correlation NMR spectroscopy has become an indispensable tool for organic chemists, serving as a cornerstone technique for the structural elucidation of a wide range of compounds [1]. Most 2D heteronuclear correlation experiments are based on 1H-13C spin pairs [1-2]; however, 1H-31P spin pairs can offer equally valuable structural information [2-4]. 31P is an NMR-active nucleus with 100% natural abundance and is present in many classes of organic compounds, ranging from ligands in organometallic catalysts to the backbone of oligonucleotides therapeutics [2-4]. Given the broad prevalence of 31P-containing compounds, it is therefore essential to employ 2D heteronuclear correlation experiments that directly probe 1H-31P spin pairs to achieve reliable structural determination and confirmation [2-4].
In contrast to 1H-13C spin pairs, the scalar couplings between directly bonded 1H-31P nuclei can be exceptionally large, reaching values of up to 600 Hz [3]. Such large 1JHP couplings pose practical challenges for heteronuclear correlation experiments that rely on INEPT-based magnetization transfer. For example, a 1JHP coupling of 600 Hz corresponds to an optimal INEPT delay of approximately 400 µs, which can be too short when a pulsed field gradient (PFG) is employed for coherence pathway selection since this limits the length and the strength of the gradient that can be applied. However, longer delays corresponding to multiples of 1JHP, can in principle be used; yet this approach generally results in reduced sensitivity due to relaxation losses [2-4]. Moreover, at extended delay times, longer-range couplings—such as 2JHP, 3JHP, and even 4JHP—may contribute to the observed correlations. Consequently, careful optimization of the INEPT delay is essential to selectively observe specific 1H-31P correlations while minimizing undesired long-range coupling artifacts [2-5].
To illustrate the application of Long-Range (LR) Heteronuclear Single Quantum Coherence (HSQC) for the detection of both directly bonded and long-range coupled 1H-31P spin pairs, a sample of 250 mM of tricyclohexylphosphine tetrafluoroborate in protonated DMSO (Figure 1) is used as a proof of concept. This compound exhibits a range of scalar couplings spanning from one-bond to four-bond interactions, making it an ideal system for demonstrating the utility and selectivity of the approach.
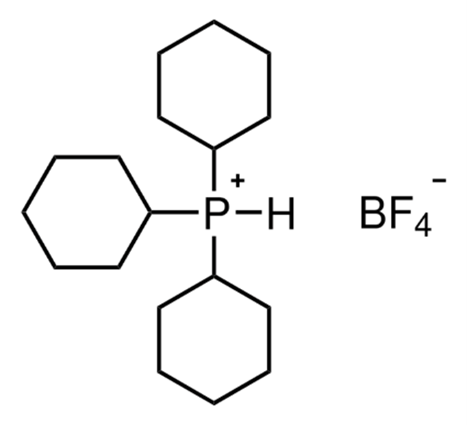
Figure 1: Molecular structure of tricyclohexylphosphine tetrafluoroborate.
NMR Results & Discussion
Two 1D 31P NMR spectra were acquired with and without 1H decoupling (Figure 2). In the absence of 1H decoupling, a broad doublet was observed with a one-bond 1H-31P coupling constant (1JHP) of approximately 400 Hz.
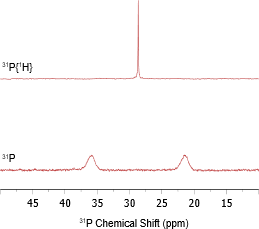
Figure 2: 1D 31P NMR spectra of tricyclohexylphosphine tetrafluoroborate (Top) with 1H decoupling and (Bottom) without 1H decoupling.
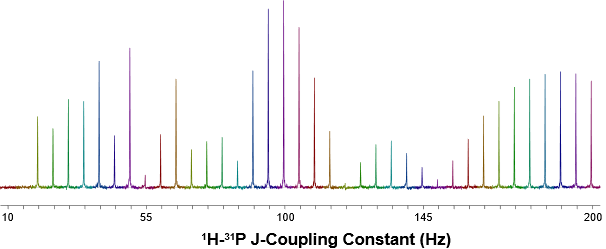
Figure 3: A series of 1D 31P refocused INEPT NMR spectra acquired with delays corresponding to different 1H-31P J-coupling constants.
To optimize the JHP coupling values for the LR-HSQC experiment, a series of 1D 31P{1H} refocused INEPT spectra, shown in Figure 3, were recorded using different INEPT delays corresponding to increasing 1H-31P J-coupling constants. The signal intensity oscillated as a function of the selected 1H-31P J-coupling values. Maximum signal intensity was achieved when the 1H-31P J-coupling constant was set to approximately 100 Hz. This optimized coupling constant reflects not only directly bonded 1H-31P spin pairs but also contributions from longer-range 1H–31P couplings. Using this value, a 1H-31P LR-HSQC spectrum was collected (Figure 4A). Technically we set the inept delays not the J-couplings. Is a very small detail but I would inverst the way it is written.
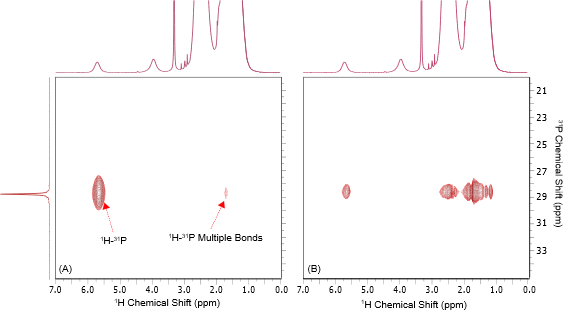
Figure 4:1H-31P HSQC NMR spectra of tricyclohexylphosphine tetrafluoroborate in protonated DMSO acquired using a 1H-31P J-coupling constant of 100 Hz (A) and 30 Hz (B). Frequency Discrimination in the indirect dimension was achieved using an Echo/Anti-echo protocol. Both spectra were acquired using 4 scans and 128 complex points with a 1s repetition delay. The horizontal trace is a 1D 1H{31P} NMR spectrum showing the 1H chemical shifts of the directly bonding 1H-31P spin pair.
Comparing the two spectra, the resonance from the one bond 1H-31P correlations and multiple bond 1H-31P correlations can be distinguished based on signal intensity: the signal from the one bond correlation is higher compared to that of multiple-bond correlations for larger 1H-31P coupling values.
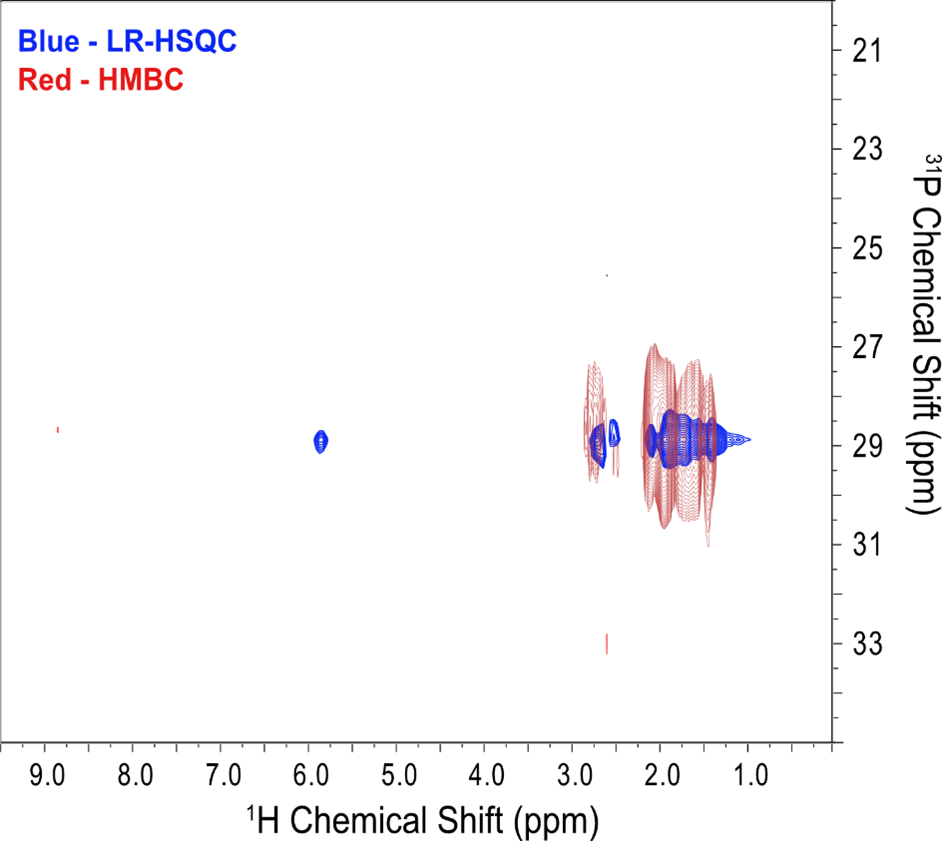
Figure 5: An overlay of 1H-31P LR-HSQC (blue) and 1H-31P HMBC (red) of 250 mM of tricyclohexylphosphine tetrafluoroborate in protonated DMSO using a 30 Hz 1H-31P J-coupling constant. [FC2.1][RS2.2]Frequency discrimination in the indirect dimension for LR-HSQC was achieved using Echo/Anti-echo protocol. Both spectra were acquired in 8 minutes.
Furthermore, when compared to a standard 1H-31P HMBC (Figure 5), the LR-HSQC offers significantly improved spectral resolution with identical experimental time and increments. In addition, LR-HSQC can resolve a one bond 1H-31P correlation without doubling the number of resonances in the 1H dimension compared to that of HMBC when the 1H dimension was acquired without 31P decoupling. Therefore, this approach allows for the unequivocal assignment of 1H chemical shifts for directly bonded 1H-31P spin pairs, offering additional structural information for 31P containing compounds.
Conclusion
1H-31P LR-HSQC provides enhanced spectral resolution for the detection of both one-bond and multiple-bond correlations, making it a valuable complement to the standard 1H-31P HMBC experiments when increased resolution is required for reliable spectral assignment. Given the structural diversity of 31P-containing compounds, 1H-31P LR-HSQC represents a powerful tool for structure determination and compound identification[2-6]. The data presented in this application note further highlights the capability of the Spinsolve spectrometers. Together with automated heteronuclear switching that enables the sequential acquisition of 1H-13C and 1H-31P 2D experiments, the Spinsolve constitutes a flexible and high-performance NMR solution for accurate spectral assignment, structural confirmation, and detailed molecular characterization.
To get the complete App Note please Click Below
References
[1] Bodenhausen, G; Freeman, R. J. Am. Chem Soc.. (1978), 100, 1, 320-321
[2] Luy, Burkard and Marino John. J. Am. Chem Soc. (2001),123,11306-11307
[3] “ NMR Spectroscopy: 31P NMR Coupling Constant ” https://organicchemistrydata.org/hansreich/resources/nmr. (access 2/28/2026
[4] Neilsen, J.T.; Arar, K.; Petersen, M.; Nuc. Acid. Res. (2006), 34, 2006-2014.
[5] Barekatain, Y. et. al. Anal. Chem. (2022), 94, 28, 10045-10053.
[6] Mao, X.; et. al.; Mol. Imaging Biol. (2013) 15, 245-249.