Diffusion PFG Gradients
All Spinsolve models can be equipped with gradient coils to generate strong pulse field gradients
Diffusion PFG Gradients
All Spinsolve models can be equipped with gradient coils to generate strong pulse field gradients
Home Products Spinsolve family Spinsolve addons Diffusion PFG Gradients
Download Resources
Besides the set of 3 D (x,y,z) pulsed field gradients for gradient enhanced methods, which is included on all Spinsolve systems, the Spinsolve can as well be equipped with a stronger pulsed field gradient for diffusion experiments.
The benefits of pulse field gradients:
- Separate the spectra of different components in a mixture by molecular size (DOSY-type experiments)
- Measure self-diffusion coefficients to understand molecular mobility (PFG diffusion measurements)
- Accelerate the acquisition of modern 2D NMR experiments (Ultra-fast 2D NMR)
DOSY (Diffusion-ordered spectroscopy)
Complex NMR spectra of mixtures can be easily separated based on the molecular self-diffusion coefficient. Consider for example the spectrum of a mixture of procaine and paracetamol in D2O. This is shown in the middle scan of Figure 1, along with the spectra of the pure compounds above and below. If we had only the mixture available, but not the pure compounds, it would be hard to figure out how many and which compounds are present in the mixture. These spectra, along with all the others shown in this post, were acquired on a Spinsolve benchtop NMR spectrometer with additional hardware to enable PFGs for measuring diffusion.
An experiment called Diffusion Ordered Spectroscopy (DOSY) creates a two-dimensional plot by acquiring a set of spectra as a function of the amplitude of the gradient. The chemical shift is measured along the horizontal axis, and along the vertical axis the self-diffusion coefficient is shown. There are tools around which perform this analysis, and one of them is the DOSY Toolbox. Using this toolbox, we have inverted the data set to create the DOSY spectrum shown in Figure 2.
What is obvious from this 2D spectrum is that the peaks are aligned along horizontal lines. Each of these lines corresponds to a different self-diffusion coefficient, and therefore a different component of the mixture. We can immediately separate the solvent peak, as well as the other two components, although their diffusion coefficients differ by a little more than 10%. This makes DOSY a very powerful tool for mixture analysis in NMR spectroscopy when the components have different diffusion coefficients.
Measurement of self-diffusion coefficients
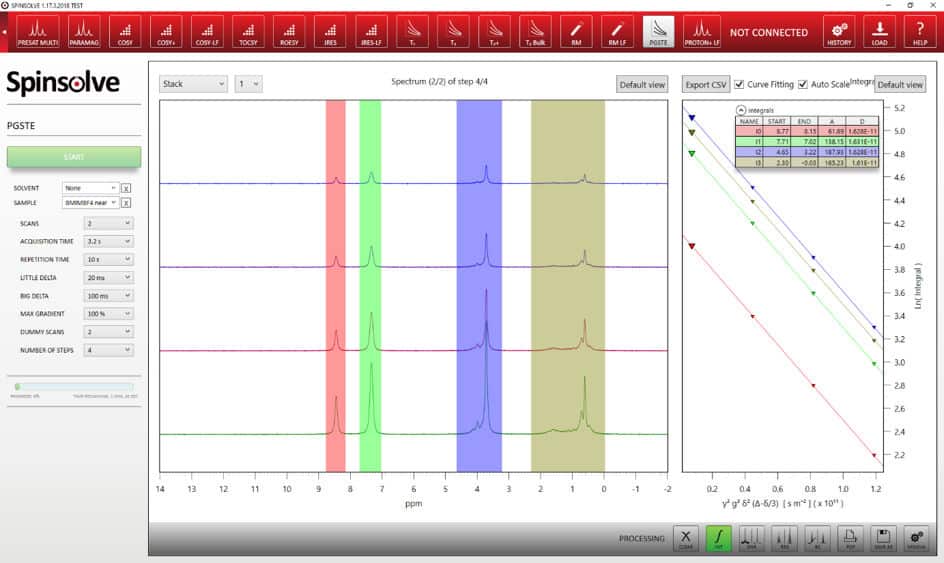
To demonstrate the possibility to determine diffusion constants, we took an example where we have three different species with three different nuclei. In this way, making use of the full power of the Spinsolve, we measure the diffusion coefficient of each nuclei to access the molecular mobility of each species in the sample. For the experiments we used mixtures of 1-butyl-3-methylimidazolium tetrafluoroborate (BMIM-BF4) and lithium tetrafluoroborate (LiBF4) at different lithium concentrations. The diffusion coefficients were measured with a stimulated – echo sequence run for each particular nuclei.
The excellent quality of the data is demonstrated by the highly linear behaviour of the individual diffusion curves. The values or the maximum gradient strength, δ and ∆ were optimized for the different samples in order to achieve a signal attenuation that covers a good dynamic range. Linear fitting of these data points gives the self-diffusion coefficients for the three different nuclei and thus the three different ions in the different samples. As expected, the diffusion coefficients decrease with increasing lithium salt concentrations due to an increase in the viscosity of the samples. Plotting the different self-diffusion coefficients as a function of the concentration of the lithium salt, a linear dependence of the diffusion coefficient is revealed, within the investigated concentration range.
Convenient software interface
- Easy user interface implemented in the standard Spinsolve® software
- Direct access to parameters related to the diffusion experiment (e.g. max. gradient strength, number of steps, δ and ∆)
- Plotted integrals and fully automated analysis done by the Spinsolve® software.
Multi-nuclear PFG spectroscopy on a benchtop NMR spectrometer
Molecular diffusion is the mechanism that defines molecular mobility in solutions. The study of diffusion processes is of interest in widespread fields in science where transport of mass needs to be determined. NMR is the method of choice to quickly determine self-diffusion coefficients of chemical species in solution. A commonly used NMR technique requires the combination of pulsed field gradients with a stimulated echo sequence (PGSTE). The spectroscopically resolved version of this sequence makes it possible to measure the diffusion coefficient of different molecules in a mixture by measuring the diffusion attenuation of the signal of each particular chemical group of each molecular structure. The diffusion version of the Spinsolve is a benchtop spectrometer that comes with gradient coils to make this type of experiments possible. The power of the combination of pulsed field gradients with spectroscopy has been demonstrated in the past to acquire DOSY (Diffusion Ordered SpectroscopY) type experiments. In this application note we take advantage of the multinuclear feature of the Spinsolve spectrometers to get access to a bigger variety of molecules that could be present in a mixture. The possibility to measure the signal of different nuclei is also advantageous in cases where signal overlapping in the 1 H spectrum does not allow for accurate separation of each component. To demonstrate the power of multi-nuclear diffusion measurements we have chosen an example where 1 H, 19F, and 7 Li provide access to three different molecular species. Read more …