716. Minimizing Ionic Interference: An APCI-MS Strategy for Real-Time Reaction Monitoring in Ionic Liquids
Chiara Salvitti, Alessia Di Noi, Francesca Cosentino, Cinzia Michenzi, Federico Pepi, Marta Managò, Isabella Chiarotto, Anna Troiani, JMassSpectrometry, (2025), DOI: 10.1002/jms.5176
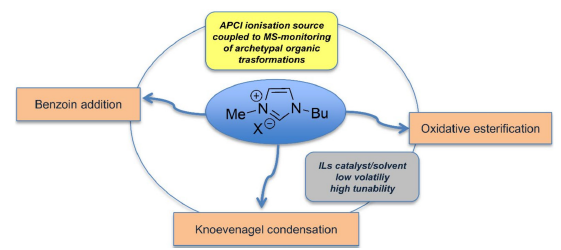
Ionic liquids (ILs) are a class of organic salts with melting points below 100°C. Owing to their unique chemical and physical properties, they are used as solvents and catalysts in various chemical transformations, progressively replacing common volatile organic solvents (VOCs) in green synthetic applications. However, their intrinsic ionic nature can restrict the use of mass spectrometric techniques to monitor the time progress of a reaction occurring in an IL medium, thus preventing one from following the formation of the reaction products or intercepting the reaction intermediates. The intense ionic signals related to the IL cation, anion, and their aggregates can indeed suppress the ionic intensities of substrates that are not intrinsically charged and often poorly ionizable. In this paper, we developed an atmospheric pressure chemical ionization mass spectrometry (APCI-MS) approach to overcome this limitation and minimize interference from the IL. The possibility of obtaining an estimation of the product yields by directly sampling the reaction mixture without quenching the process was tested by constructing calibration curves on three different model reactions, namely, (i) Knoevenagel condensation, (ii) oxidative esterification of aromatic aldehydes, and (iii) benzoin addition. A good correlation was obtained between the product yields measured by the APCI-MS procedure and those extrapolated from the isolated products. These results highlight the potential of this approach for real-time monitoring of reactions in ILs, eliminating the need for time-consuming extraction steps and minimizing the risk of losing critical mechanistic information.