740. Determination of Fe(II) and Fe(III) in Paramagnetic Matrices Using NMR Relaxometry
Kaixu Jin, Caipeng Xu, Yufen Zhao, Songsen Fu, AnalyticalChemistry, (2025), 10.1021/acs.analchem.5c02587
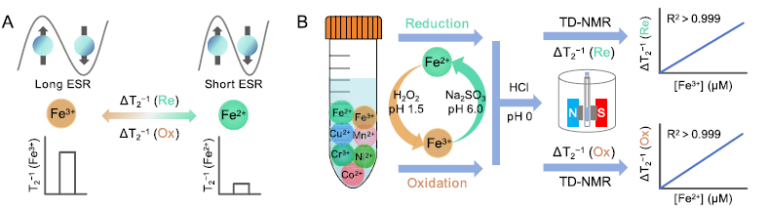
Accurately determining the concentration and redox ratio of iron ions is crucial in fields such as biogeochemistry and environmental chemistry. While current NMR relaxometry-based methods are lauded for their simplicity and speed, they face challenges when paramagnetic matrices are present. This study overcomes this limitation by leveraging the redox sensitivity of iron ions. By using appropriate redox reagents and pH conditions, seamless transitions between Fe2+ and Fe3+ are achieved without altering the valence states of other common paramagnetic metals. The significant difference in the relaxometry rates of water protons induced by Fe2+ and Fe3+ enables a strong linear relationship across a concentration range of 0 to 500 μM, with a detection limit of less than 1 μM. The established standard curves were effectively applied to determine the redox ratio and total iron concentration in both artificial and real samples, validated by X-ray photoelectron spectroscopy (XPS) and inductively coupled plasma-optical emission spectrometry (ICP-OES) analyses. Furthermore, the method demonstrates resistance to interference from common ligands, suggesting its promising potential for analyzing biological and food samples.