743. Design, synthesis, and evaluation of anticancer activities of 1,2-diborolane derivatives for hepatocellular carcinoma: An in vitro and in silico study
Yuksel Sahin, Gizem Antika, Cagdas Aktan, Kubilay Metin, Huseyin Ozgener, JMolStruc, (2025), 10.1016/j.molstruc.2025.144690
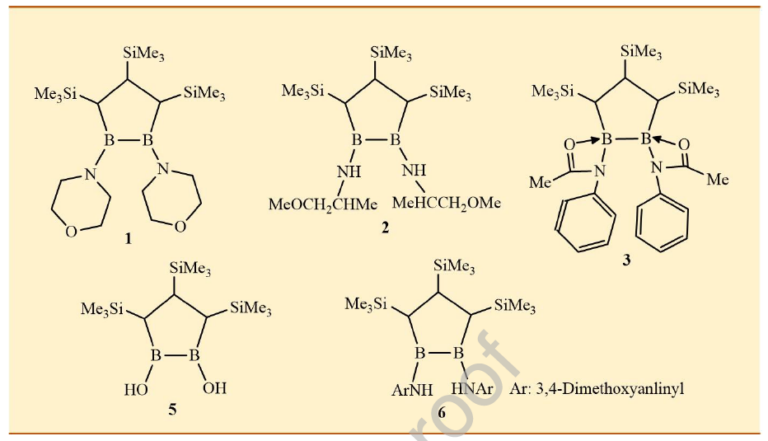
Hepatocellular carcinoma (HCC) is the most prevalent form of primary liver cancer and remains a major global health challenge due to limited treatment options and poor prognosis. Boron containing compounds have garnered attention for their diverse biological activities, including pro-apoptotic effects in various types of cancer. In this study, we synthesized a panel of novel 1,2-N-substituted-1,2-diborolane derivatives and evaluated their antiproliferative, antimigratory, and apoptotic effects on hepatocellular carcinoma cell lines, HepG2 and Hep3B. Spectroscopic analyses confirmed the structural integrity of the synthesized compounds, revealing characteristic ¹H-, ¹¹B-, and ¹³C-NMR shifts consistent with boron-oxygen and boron nitrogen bonding patterns. The derivatives, particularly compounds 2, 3, and 6, demonstrated potent and selective cytotoxicity toward HCC cells, with compound 3 exhibiting the lowest IC50 value (6.75 μM) in HepG2 cells. Their time-dependent anti-proliferative effects were further supported by colony formation assays demonstrating long-term growth suppression, while wound healing assays revealed marked inhibition of HepG2 cell migration, indicating the compound's anti-metastatic potential. Our results demonstrate that the compound significantly induces apoptosis, modulates the expression of key apoptotic genes (Bax, Bcl-2, and caspase-3). In silico molecular docking further confirmed strong binding affinity to the anti-apoptotic Bcl-2 protein, supporting the proposed mechanism of action. These findings highlight the compound as a promising candidate for further preclinical evaluation in liver cancer therapy.